One of the Top 5 Wildfire Smoke Hazards is Carbon Monoxide (CO), present at different stages in wildland firefighting operations.
How is CO produced?
Carbon Monoxide is produced by incomplete combustion of wood or other organic materials, due to lack of air to burn. All fuel need a certain amount of oxygen to burn completely through the chemical oxidation reaction that causes fire and if this does not happen, this colorless, odorless, inaudible and highly toxic gas is generated.

Picture from: Pixabay – skeeze
Why is Carbon Monoxide toxic?
Carbon monoxide has a much higher affinity for hemoglobin than oxygen, reacting with it and forming the Carboxyhemoglobin ((COHb). Once COHb is formed, it cannot longer capture the oxygen, mainly affecting two of the most important organs, the brain and the heart that require large amounts of oxygen to function properly.
What are the immediate effects?
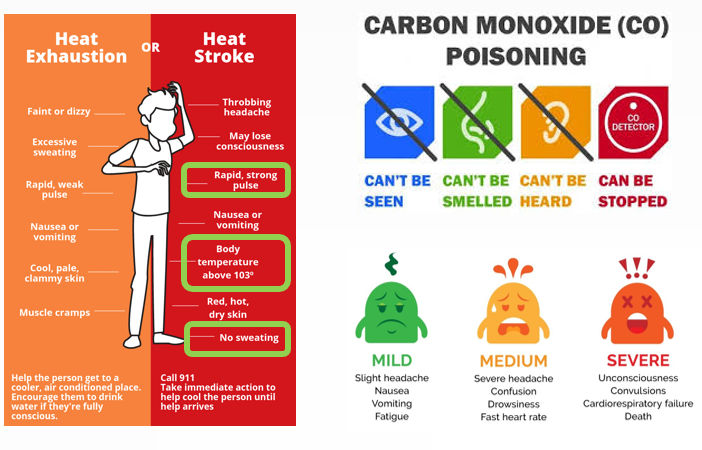
The inhalation of the carbon monoxide causes severe health effects almost immediately, ranging from diminished work capacity and a loss of visual perception, manual dexterity, driving performance, and attention level, slight headache and nausea (at mild exposures) severe headaches, dizziness, loss of perception of the environment, tachycardia and tunnel vision (at medium exposures), severe headaches, dizziness, loss of perception of the environment, tachycardia and tunnel vision (at medium exposures), loss consciousness, seizures, heart failure or death (at high exposures).
At very high levels in very close proximity to a smoldering fire, intoxication by CO can trigger an angina attack, increase abnormal heartbeats, and potentially lead to sudden heart failure in individuals suffering from preexisting heart diseases,.
This infographic might be used as an indication guide to detect and evaluate the degree of intoxication.
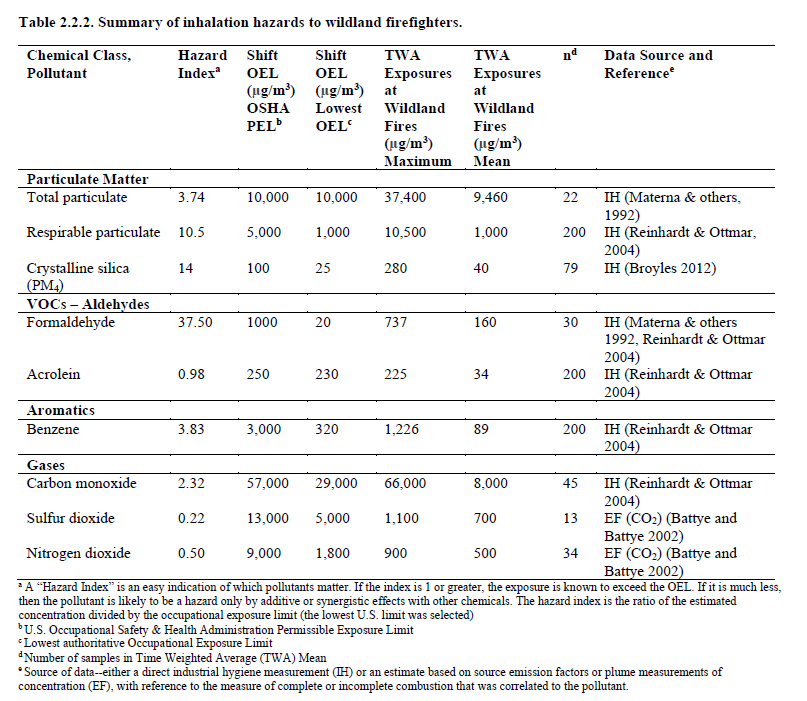
How much exposure do wildland firefighters get?
The Occupational Safety and Health Administration (OSHA, the main federal U.S. Agency in charge of enforcing safety and health legislation), established a framework, by analyzing the main pollutants in forest fire smoke and by drawing a maximum exposure index (OEL). It is common to find that firefighters are exposed to too much CO (5 to 10 percent of the time over the course of an average work shift).
Last November, The National Wildfire Coordination Group published the "Smoke Management Guide for Prescribed Fire". The investigations they carried out established that “When people are exposed to CO, the time until they reach a toxic level of COHb can be predicted as a function of CO concentration, breathing rate, altitude, and other factors. The harder the work and the higher the altitude, the more rapidly COHb forms at a given concentration of CO. In heavy smoke where there is a high level of CO, symptoms of overexposure to CO can occur during hard physical labor after 15 minutes.”
Apart from Carbon Monoxide, wildland firefighters are exposed to organic and inorganic gases and vapours, acids, amines, formaldehyde, acrolein, NOx and VOCs. In Urban areas, the additive or synergistic effects of vegetable fuels with other chemicals increase the pre-existing risk.

Picture from: Greenpeace.
How can we eliminate CO?
Carbon monoxide is not a gas that can be filtered with activated carbon, it must be eliminated with a catalyst, as we see in the automotive or mining industries.
What solutions are available in the market?
Until now, no multi-gas filter was capable of removing CO. The only possible solution to protect firefighters from CO was the use of an ARE (Autonomous Breathing Equipment). These are respiratory systems with compressed air bottles non recommended for wildland firefighting operations due to their weight and size.
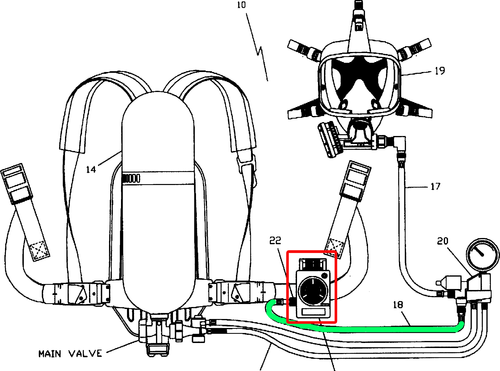
New vft Astrea filter, the first and only filter in small size and long lasting, able to protect against CO, especially designed for wildland fires
Astrea Materials and Vallfirest have developed a CO filter able to efficiently protect wildland firefighters in firefighting operations and prescribed burns. It is the first and only filter for Carbon Monoxide, Formaldehyde, Nitrogen Oxides and other toxic compounds present in wildland fires. An exclusive and patented gold nanoparticulate formula, resulting from 15 years of basic research and industrial development.

Picture from: Vallfirest.
How does it work?
The product is composed of a patented precious metal catalyst and an additional P3 R particle filter. Gold nanoparticles have the unprecedented ability to eliminate contaminating compounds such as CO, NOx and formaldehyde at room temperature and in the presence of humidity (>90 % RRHH) without the need of any desiccant stage, as would otherwise be required with conventional materials such as hopcalite. The result is a product that offers stable removal of airborne pollutants for long periods of time and therefore becomes of practical use in wildland fire extinguishing work. The cartridge is completed with a certified P3/P100
Its nanostructured catalyst can remove the toxic compounds in a wildland fire with a 98% of filtration efficiency.
How do we connect it?
Bayonet for direct connection to the Xtreme Mask, guaranteeing a good seal and comfortable use in wildfire, compatible with all types of glasses, hearing protectors and with most helmets available on the market.

The half mask of the Xtreme Mask has a low nasal profile (which it offers a perfect sealing on Caucasian, Asian, African-American and American faces), and it has two exhalation valves (so that carbon dioxide escapes faster, increasing respiratory comfort). The textile mask is certified according to EN 15614: 2007, offering high protection against radiation and incandescent particles.
Fire test results
The CO filter was tested on the field. These are the data resultant after several evaluations.
In typical shift of 6 h:
- CO Filtration > 98%
- NOx Filtration >98%
- Formaldehyde removal (formaldehyde is turned to CO2 and H2O) > 98%
- Additional protection from VOCs
For more information, don't miss this video: